Monkeypox vaccine in the US: What public health experts know so far—and what they don't
Monkeypox vaccine in the US: What public health experts know so far—and what they don't
The current supply of monkeypox vaccine in the U.S. is limited, but the federal government has released a national vaccine strategy to be undertaken by the Food and Drug Administration, Centers for Disease Control and Prevention, National Institutes of Health, and the Administration for Strategic Preparedness and Response.
In advance of vaccines becoming more widely available, there are actions people can take to mitigate their risk of exposure and infection—actions that are also useful even between the first and second shot for those who have qualified for vaccination.
While monkeypox has not been classified as a sexually transmitted disease, it has been shown to be highly contagious in the context of close, intimate human contact. Therefore, exchanging contact information with any new partner to allow for sexual health follow-up, speaking openly with your partner about any monkeypox symptoms, and avoiding sharing things such as towels, sex toys, and toothbrushes are means by which people can proactively try to protect themselves from exposure. Close social situations, such as parties, raves, or nightclubs where patrons wear minimal clothing and where there is often direct bodily contact, also increase a person's risk.
Stacker compiled a list of frequently asked questions about the monkeypox vaccine from governments, scientific sources, and health experts. Stacker spoke with Dr. Wafaa El-Sadr, professor of epidemiology and medicine and chair of global health at Columbia University's Mailman School of Public Health, and Dr. Sandra A. Fryhofer, board-certified in internal medicine and chair of the American Medical Association's Board of Trustees, to gain their expert perspectives on the current state of vaccine dissemination and to discover what the near future holds for increased vaccine availability.
As of Aug. 30, 2022, there were 18,417 total confirmed monkeypox cases in the United States, according to the CDC.
What types of vaccines are available in the US?
The U.S. government has two stockpiled vaccines against monkeypox—Jynneos and ACAM2000. As of this writing, the Jynneos vaccine is the only one approved for public distribution.
Dr. Fryhofer clarified why ACAM2000 is not being administered. "It is a percutaneous vaccine and that makes it more challenging to administer and it is riskier, and therefore, you need more trained personnel," she told Stacker.

Who is eligible to get it?
Each state has its own recommendations, but the CDC recommends vaccination for people who have been exposed to monkeypox and people who may be more likely to get monkeypox, and these include:
- People who have been identified by public health officials as a contact of someone with monkeypox
- People who know one of their sexual partners in the past 2 weeks has been diagnosed with monkeypox
- People who had multiple sexual partners in the past 2 weeks in an area with known monkeypox
"The vaccine is being prioritized for post-exposure, and those with certain risk factors," explained Fryhofer, who added that the reason why not everyone is being called for vaccination is that there are not enough vaccines immediately available. Fryhofer also said that the vaccine may reduce symptoms but not prevent them.

How can someone who qualifies get the vaccine?
If you are eligible, make an appointment in one of your city or state vaccination locations, either online or by phone. Use a simple Google search with the format "Appointment monkeypox vaccine (state, city, or county)" to find out how to get an appointment.
Dr. El-Sadr told Stacker, "We should try to make vaccines available to people who need them as soon as possible, and also assure people that this vaccine has been shown to be safe and effective."

Who cannot get vaccinated?
The New York City Health Department fact sheet about the Jynneos vaccine states that people who should avoid the vaccine include those who have had a severe allergic reaction to a prior dose or an ingredient in the vaccine, such as gentamicin, ciprofloxacin, or egg protein. But groups that might otherwise need to take caution on a novel vaccine—including pregnant or breastfeeding people, people with a heart condition or HIV, or people with a weakened immune system or skin condition—can get the vaccine.

Why is it important to get it?
"I think it's important for people to get the vaccine if they are in danger," El-Sadr said. She went on to explain that the danger groups are quite specific: Individuals with close contact to someone diagnosed, because the vaccine may prevent them from getting the infection; people at high risk, such as men who have sex with men, as most of the infections are occurring within this group; and finally, people who are at risk of developing severe monkeypox, such as those living with HIV or AIDS.
How many doses are required?
The Jynneos vaccine—again, the only vaccine approved for use by the FDA at this time—requires two doses administered 28 days (or about four weeks) apart.
ACAM2000 is a single-dose vaccine, and it takes four weeks after vaccination for its immune protection to reach its maximum. However, it has the potential for more side effects and adverse events than Jynneos, and this is why it is not recommended for people with severely weakened immune systems and several other conditions.
Are there any side effects?
While side effects are a potential reality for any administered medication, El-Sadr explained that "Jynneos has well-tolerated effects—which means it causes few side effects, the most common one being pain where the vaccine was administered. So, only local side effects. It might also cause more general side effects, like itchiness, fatigue, or headache, but that is rare."
ACAM2000 has the potential for more side effects and adverse events than Jynneos. This is why it is not recommended for people with severely weakened immune systems and several other conditions. More about each vaccine's side effects can be found at the FDA website.
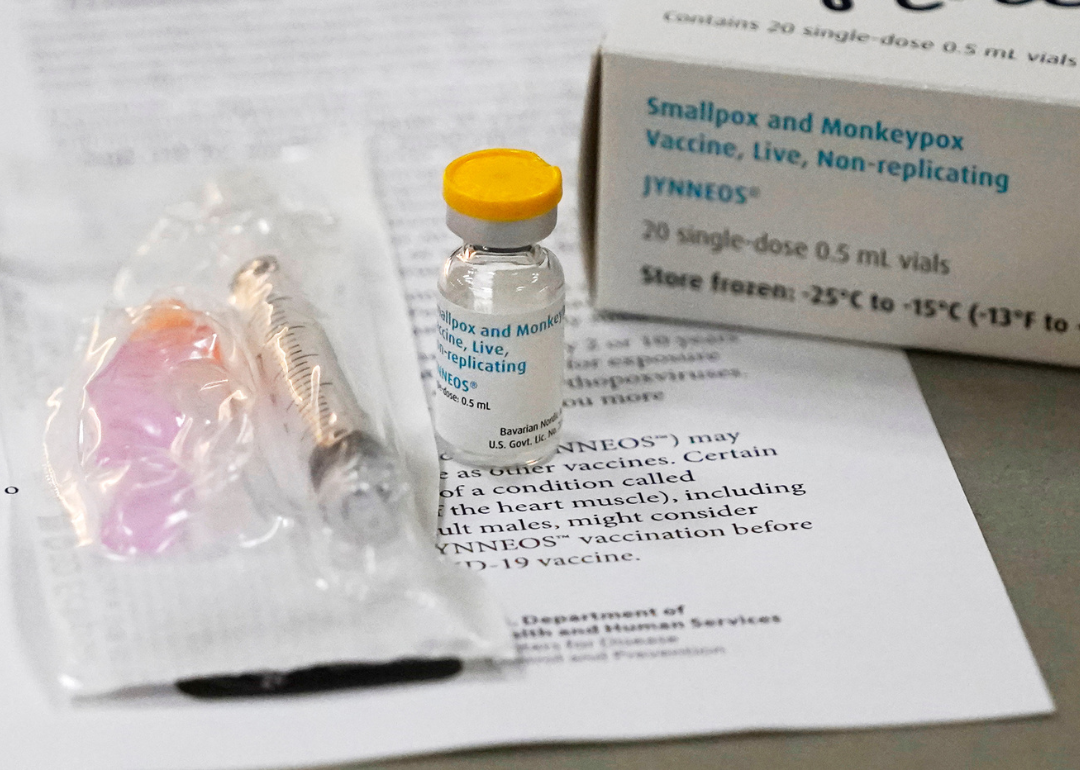
Can you get monkeypox or smallpox from the vaccine?
The vaccine cannot cause monkeypox or smallpox in the vaccinated individual or those near them. This is because the virus in the vaccine is weakened and cannot copy into human cells.
"The monkeypox vaccine is developed for smallpox as well as monkeypox," El-Sadr explained. "It essentially contains a type of pox that contains a virus from the same family as monkeypox and smallpox, but it's not able to multiply or replicate in a person."

How effective is the vaccine and when is a person considered vaccinated?
The Jynneos vaccine's maximum immunity is 14 days after the second dose. With ACAM2000, it takes four weeks after vaccination to be fully protected.
According to the American Medical Association, both vaccines are believed to be about 85% effective at preventing monkeypox, but to date, neither has been studied for post-exposure prevention. "We do not have information on vaccine effectiveness for monkeypox in this specific context," Dr. Caroline Schrodt, an expert from the CDC's Division of High-Consequence Pathogens and Pathology in Atlanta, told the AMA.
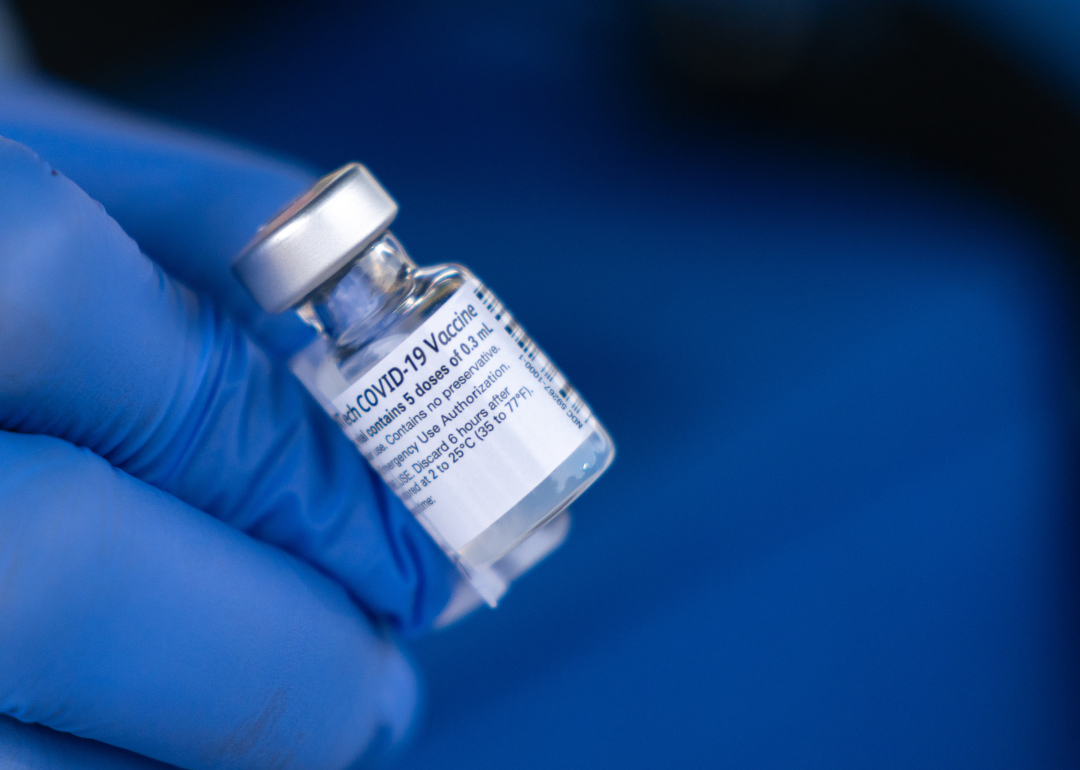
How is it similar to or different from the COVID-19 vaccines?
As Fryhofer explained, "these vaccines have been available for years—ACAM2000 since 2007 and JYNNEOS since 2019. There are no new vaccines already studied and available. We need to learn from our mistakes with COVID and educate the public about symptoms, what to look for and what to do, disposition [toward infection], and how to avoid monkeypox, so we can stop the spread."
"Also, we need to push the government to get more doses than [what] we have, because we have funding to stop the spread," Fryhofer added. "But we have to act quickly and improve the health of the nation. We could be really getting into another pandemic if monkeypox is spread everywhere around schools, health care facilities, and elder care facilities."
El-Sadr said there are differences and similarities between the two types of vaccines. "There are many different kinds of COVID vaccines," she pointed out. "We have two vaccines that are messengerRNA or MRNA—Pfizer and Moderna. The other is made from protein; this one contains a weakened form of the virus. Moderna and Pfizer don't contain the virus itself. The immune system will [instead] produce some portion of the virus that will generate protective antibodies against the virus."
Does the chickenpox vaccine offer any immunity to monkeypox?
The chickenpox vaccine, or a previous infection of chickenpox, does not offer any protection for monkeypox, according to the CDC.

What if someone has just had monkeypox or suspects they might have had it? Should they also get vaccinated?
El-Sadr was firm: "No, you should not vaccinate if you have been diagnosed or suspected of having monkeypox." Her suggestion is to go and seek care "so that you can be examined and evaluated, and hopefully then the individual will be either diagnosed with monkeypox or not."
If you have had the infection already, El-Sadr's advice is to "isolate, call your close contacts to let them know, and you don't need to get vaccinated."
What do we still not know?
According to the FDA, any conclusions as to whether any dosage or sequence of dosages beyond the presently approved two doses of Jynneos may provide adequate protection against monkeypox is still pending. For now, the only currently approved regimen is the two-dose regimen.

Where can I find trustworthy information about the monkeypox vaccine?
Make sure you are looking at official websites, with science-based information. The following sites are reputable sources:



